15 7 月 雙盲隨機對照研究為生物共振療效提供科學實證

雙盲隨機對照研究為生物共振療效提供科學實證
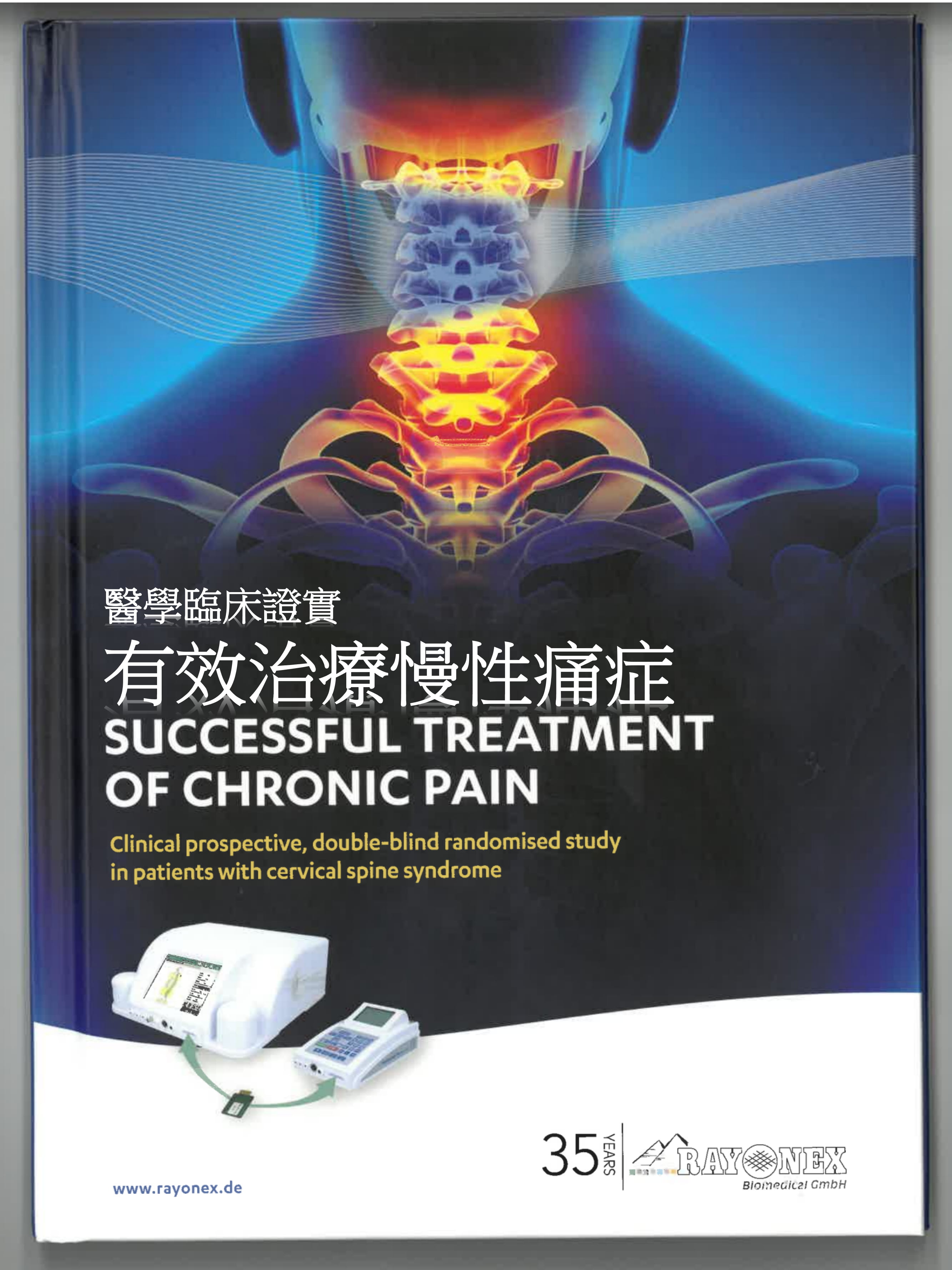
在德國完成的最新一個雙盲隨機對照實驗,證實了 Rayonex 生物共振療法的效用與安全性。該實驗的對象是頸椎綜合症患者,這是一個影響約兩成人口的痛症疾病。參加試驗的病人共 54 名,全部患有起碼中度的頸椎痛症超過三年,他們被分為實驗組和對照組又稱控制組),分別接受真實的或仿擬的生物共振治療。
頸椎綜合症的徵狀頗為廣泛,包括頸椎、肩膀、手臂和關節痛、喪失活動機能及神經痛,有時亦會感到頭痛、暈眩、視力模糊及耳鳴等。
經過 10 次每次 130 分鐘的治療後,接受了生物共振療程的實驗組病人表示在頸痛、頭痛、肩膀痛或肌肉緊張方面有顯著的改善,而只接受了安慰劑療程的對照組病人則表示沒有明顯改善。
在日常活動方面,實驗組病人表示在運動、行山、家務、園務、游泳、騎單車及社交活動上都有顯著的改善,而對照組則表示沒有。
在痛楚以及身心健康方面的感受與感觀上,實驗組都表示有明顯改善。
總括而言,實驗組在客觀和主觀方面的指標都達到了明顯的進步,並表示會將生物共振療法推薦於他人。
這臨床研究在嚴格監控下實行,所得出的正面結果,為多年來治療師與病人的見證加以肯定,亦為早前多項觀察性研究的結論帶來科學實證。
此研究用的儀器是 Rayonex PS1000 4.0 Polar 生物共振機,是歐盟認證許可的醫療儀器。
生物共振療法用和諧共振的頻率為身體增加能量促進正常運作,是自然療法中的一種非入侵性方法。今次治療頸椎綜合症的頻率程式由專業治療師設定,旨在增加能量、提升體內平衡,並改善病症、排毒、疼痛以及心理壓力。研究的正面結果充分肯定了程式的療效。
這是 Rayonex 生物共振儀器第一次被用於雙盲隨機對照實驗,這種實驗被視為最高標準,最科學化的研究,能提供一個治療有效與否的實證。
參加這個研究的病人被隨機分為實驗組和對照組,實驗組接受真實由儀器運作的生物共振治療,而對照組則接受由同樣儀器運作,但沒有被真實啟動的安慰劑「治療」,病人本身以及前線研究員都並不知識各病人的組別。這個做法是為了避免結果受安慰劑效應或觀察者偏向所影響。由於病人本身也不知道自己是否真的接受了治療,他們也不會太輕易表示治療有效。
這個實驗的參加者包括 17 位男士與 37 位女士,年齡介乎 28 與 84,他們都清楚知道這個研究為探討 Rayonex 生物共振療法對頸椎綜合症的效用。挑選他們的標準包括年齡超過 18 歲、頸椎痛的「頸殘疾指數」達至起碼中等、他們有能力及意願去回答問卷以及提交知情同意書。
所有病人都確認他們持續患有頸椎綜合症三年以上,並同意在研究的開始和完結之前透過「頸殘疾指數」和「視覺模擬量表」確認疼痛程度。
實驗與對照兩組的參加者都包括相若比例的患有其他病症的人士,在實驗期間他們全部都繼續服用他們慣常的藥物,並依循他們一貫的餐飲與生活習慣。
該研究不被納入的標準包括患有威脅生命的疾病、炎性骨骼肌疾病、骨折或手術後創傷、嚴重影響活動能力的退化疾病、懷孕或哺乳中、精神病、酗酒或吸毒、喪失能力、被監禁,以及受僱於或依賴贊助此研究的人士,還有正在參加其他研究的人士。
此實驗經過德國下薩克森州倫理委員會考核,確認並不抵觸任何道德標準。
此實驗長達 12 星期 (包括治療後問卷訪問的時間),第一位病人在 2019年5月27日 參加而最後一位病人在 2019年11月12日 完成。在此期間,有共 7 位參加者因各種私人原因退出而沒有完成實驗,所以最終被統計的人數是 24 位實驗組對象以及 23 位對照組對象,共 47人。
2020年2月,作者 Hans Werner 醫生完成了題為「隨機、前瞻性、雙盲研究:證實 Rayonex 生物共振儀對治療頸椎綜合症安全而有效」的報告,他作出以下結論:
「相對安慰劑儀器,PS1000 Polar 4.0 生物共振儀在所有客觀及主觀的標準上都達到顯著的病況改善效果。此研究尤為重要因為它採用了安慰劑儀器去達到雙盲的設計效果。
此研究更證實了 PS1000 Polar 4.0 生物共振儀的安全性…. 在研究期間沒有出現任何可改變儀器風險狀況的不良效果。」
Rayonex 生物醫學公司設於 Lennestadt,由工程師 Paul Schmidt 在 38 年前創立,依循他所應用的生物共振原理製成醫療儀器,獲得德國有關機構每年核實及發出歐盟許可認證,並銷售於 45 個國家。
Double-blind Randomised Controlled Study gives Scientific Proof to Bioresonance Therapy
A new clinical study in Germany has given fresh proof to the safety and effectiveness of bioresonance treatments by Rayonex medical devices. The study was a prospective, double-blind, randomised study involving patients with cervical spine syndrome (CSS), a pain condition that affects some 20% of the population. The study of 54 patients, half assigned to the treatment group and half the placebo control group, was aimed at determining whether bioresonance treatments by Rayonex medical devices could alleviate pain and restricted movements associated with CSS. All patients in the study had at least moderate pain in the cervical spine region for over 3 years.
Cervical spine syndrome has relatively non-specific symptoms. It can cause pain, dysfunction and nerve irritation in the neck, shoulders, arms and joints. These may be followed by headaches, dizziness, visual disorders and tinnitus.
After 10 weeks, patients who received weekly treatments (130 minutes each time) from a Rayonex bioresonance device reported significant improvements in neck pain, headaches, back pain, shoulder pain or muscle tension. Those in the control group who received placebo treatments reported no significant improvements.
In terms of physical capacity, patients in the treatment group reported significant improvements in parameters including sports, hiking, housework, gardening, swimming, biking, everyday life and social activities. Those in the placebo control group reported no significant improvements in the same activities.
In terms of pain, vitality, general health perceptions, physical functioning, social functioning, emotional health and mental health, treatment patients reported significant improvements in all aspects. Those in the control group reported no significant improvements.
In sum, the group that received bioresonance treatments achieved significant improvements in all objective and subjective parameters compared to the control group. All patients in the treatment group said they would recommend the therapy to others, while only around a quarter of patients in the placebo group said the same.
The positive results from study – carried out under the strictest scientific protocols – support positive findings from previous observational studies as well as submissions by therapists and patients over many years.
The device used for the study is the Rayocomp PS 1000 Polar 4.0 bioresonance device. It is an EU certified / authorised medical device under the Medical Devices Act.
Bioresonance therapy, which uses harmonious resonating frequencies to energise the body’s normal regulatory mechanisms, is a non-invasive treatment method in naturopathy.
The frequency patterns used for treatments in this study target energy supply, physiological imbalances, pathological symptoms, detoxification, pain and psychic stress. They are put together into programs by experienced practitioners. The positive results of the study validate the therapeutic effects of the programs.
It is the first time that the efficacy and safety of a Rayonex bioresonance device has been subjected to a double-blind randomised controlled study. Such a study is considered to be the “gold standard” of epidemiological studies as they provide the strongest possible scientific evidence of therapeutic effect.
In the present study, subjects were assigned at random to intervention (in this case bioresonance treatment) and placebo control (in this case inactivated devices which provide the appearance of treatments but produce no effect). Neither the subjects nor the investigators knew who were receiving real treatments. This was supposed to eliminate any subjective, “placebo” feeling of benefit due to a belief in treatments. Participants would also be deterred from over-zealous reporting of positive results since they did not know whether they were in the treatment group or the control group.
For this study, the participants were 17 men and 37 women aged between 28 and 84. They were recruited for the specific purpose of testing the efficacy of the Rayonex bioresonance device for treating CSS. The inclusion criteria included age (at least 18), pain in the cervical spine region of at least a moderate degree on a “Neck Disability Index”, ability and willingness to answer questionnaires and provide written informed consent.
All the patients confirmed that they had been diagnosed with CSS for at least 3 years, and agreed to be assessed by way of a Neck Disability Index and a Visual Analogue Scale before and after treatments. The Neck Disability Index is an established, validated measuring instrument for quantifying the pain caused by CSS, and the Visual Analogue Scale has been used to record pain intensity as an indicator of significant symptoms.
Both treatment and placebo groups had a similar proportion of patients with various pre-existing medical conditions typical of population samples. All patients continued with their pre-existing medication regime as well as their customary diet and lifestyle.
The study had exclusion criteria encompassing systematic life-threatening diseases, inflammatory musculoskeletal diseases, trauma from fractures and surgeries, severe degenerative diseases with significant restricted movements, pregnant or breast-feeding women, mental illnesses, alcohol or drug abuse, incapacitation, incarceration, people employed by or were dependent on the sponsor or investigator, and people taking part in another study.
There was no ethics issue and the Lower Saxony ethics committee voted in favour of carrying out the study.
The study lasted 12 weeks including interviews. The first patient was included on 27/05/2019 and the last patient finished participating on 12/11/2019. During the study there were 7 patients who dropped out due to various personal reasons. The final number of subjects were thus 24 in the treatment group and 23 in the placebo group, making a total of 47.
In the final clinical report of Feb 2020, titled “Randomised, prospective, double-blind study to provide objective proof of the performance and safety of the Rayocomp bioresonance device in patients with cervical spine syndrome”, the author Dr Hans Werner gave the following conclusion:
“The Rayocomp PS 1000 polar 4.0 bioresonance device achieved significant improvements in all objective and subjective parameters compared to the placebo device. This study is particularly important as it adopts a double-blind design involving a placebo device for comparison.
“Furthermore, the study confirms that the Rayocomp PS 1000 polar 4.0 bioresonance device is safe…. No adverse effects were detected during this clinical trial that could alter the risk profile of the Rayocomp PS 1000 polar 4.0 bioresonance device. The favourable safety profile of the medical device was confirmed.”
Rayonex Biomedical GmbH was established in Lennestadt 38 years ago by engineer Paul Schmidt, who found a unique way to harness frequencies for healing and harmonisation. Following his approach Rayonex developed machines that are annually audited and certified as EU approved medical devices, and has been distributing them in 45 countries.

Sorry, the comment form is closed at this time.